|
4/17/2024 0 Comments Density of water gcm = g/cc = g/ml = g/mL - they are all the same. (rho 1.66 gcm-3) How to Find Volume with Density and Mass: You can readily find the volume of an object by rearranging the density equation. This structure consists of two hydrogen atomsĭivided by 16.02 = lbs/cu.ft. The density of water is equal to (1) gram per cubic centimeter The earth density is equal to (5.51) grams per cubic centimeter. Thus 1 g water will have 6.022×1023 18 molecules H 2 O 0.334 × 10 23 molecules H 2 O. Now 18 g water contain 6.022 × 10 23 moleculesH 2 O. Number of moles of water in 1g 1 18 moles 0.055 moles H 2 O. Maximum density does not occur when solidified. The density of water is approximately 1g/ml. Fresh water has a maximumĭensity at around 4° Celsius. It expands rapidly adding about 9 % by volume. Substance on Earth that exists in all three physical states of matter: The density of water varies with temperature and impurities. % of the Earth's surface is covered with about 1.36 billion cubic kilometersĭensity of pure water is a constant at a particular temperature, andĭoes not depend on the size of the sample. Most animals and plants contain more than 60 % water by volume. Has a density (weight or mass) of about 1 g/cu.cm, 1 g/ml,ġ kg/liter, 1000 kg/cu.m, 1 ton/cu.m or 62.4 lb/cu.ft See other water links at the bottom of this informational page. We strongly implore you to conserve our natural resources, all of the, but especially our water. In the information below, you will find some interesting general data about water. 1\,\texty) below it.Without water, there is no life on our Earth. 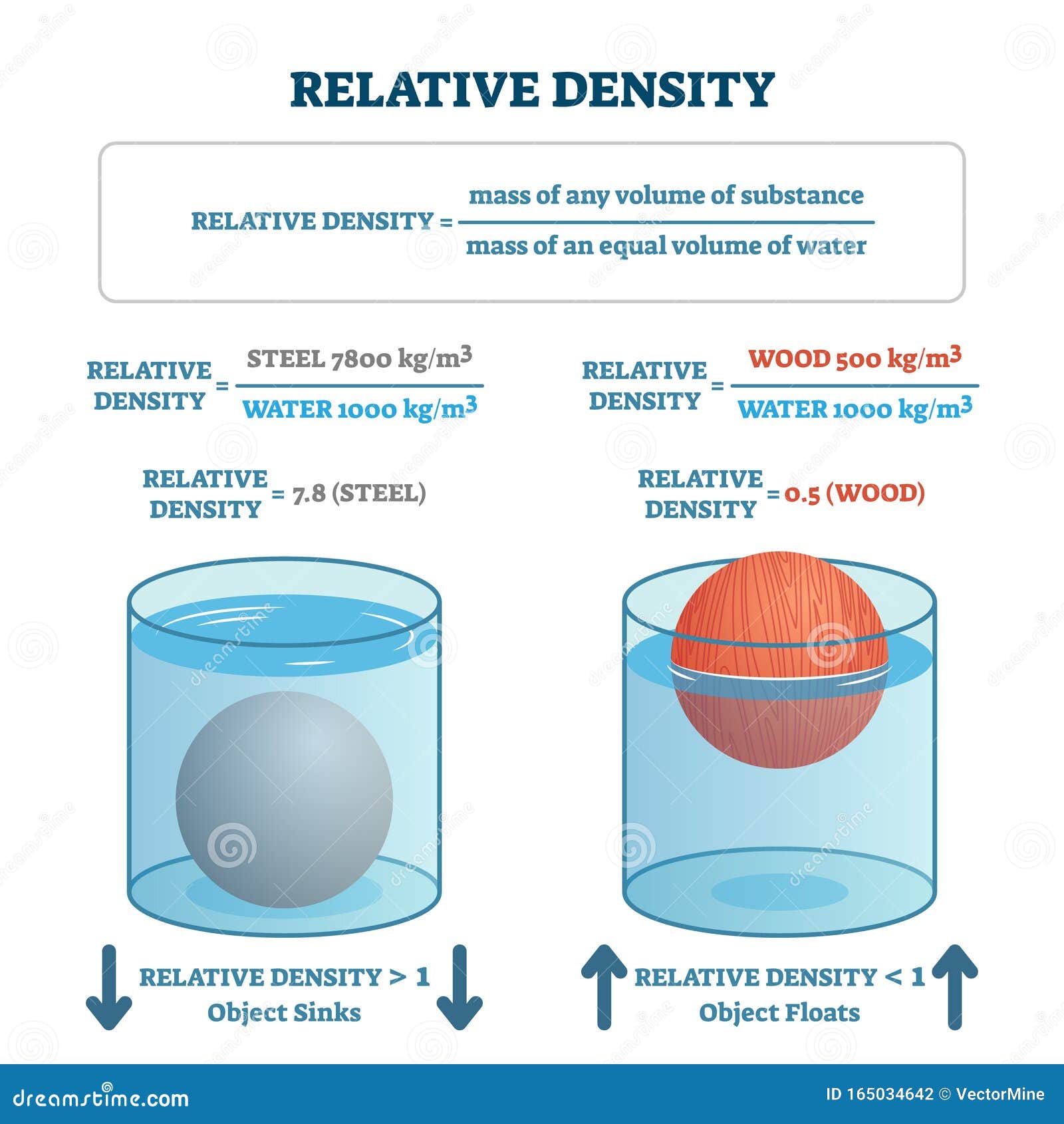
Plasma will not be discussed in depth in this chapter because plasma has very different properties from the three other common phases of matter, discussed in this chapter, due to the strong electrical forces between the charges. At high temperatures, molecules may disassociate into atoms, and atoms disassociate into electrons (with negative charges) and protons (with positive charges), forming a plasma. There exists one other phase of matter, plasma, which exists at very high temperatures. Further cooling of the surface produces less dense water, and the lake becomes stably stratified with regard to temperature-controlled density. In this chapter, we generally refer to both gases and liquids simply as fluids, making a distinction between them only when they behave differently. By the time the surface water reaches 4 ☌ (39.2 ☏), the temperature of maximum density for fresh water, the density-driven convective overturn has reached the bottom of the lake, and overturn ceases. It is exactly 1000 kg/m3 at 4 degrees Celsius. Take, g 10 m / s 2, density of water 10 3 k g m 3. Calculate the tension in the string (a) before and (b) after the metal is immersed. But you’re right that it does vary a little bit with temperature. A piece of aluminum with mass 1.00 k g and density 2700 k g / m 3 is suspended in air from a string and then completely immersed in a container of water. Rocks often have a density around 3 g/cm 3, and metals often have densities above 6 or 7 g/cm 3. If you remember that the density of water is very close to 1.0 g/ml or 1.0 oz/fluid ounce ('a pints (16 oz) a pound (16 oz) the world around'), you may notice that if a cup is 236.6 ml, as given, it should weigh 236.6 g. When placed in an open container, gases, unlike liquids, will escape. In general, we say that the density of water is 1000 kg/m3 (or 1 g/cm3). Liquids often have densities of about 1.0 g/cm 3, and indeed, fresh water has a density of 1.0 g/cm 3. The fruit has a density of 1.045 g/ml, while the syrup has a density of 1.15 g/ml, so the fruit will float, being less dense. This makes gases relatively easy to compress and allows them to flow (which makes them fluids). In contrast, atoms in gases are separated by large distances, and the forces between atoms in a gas are therefore very weak, except when the atoms collide with one another. Because the atoms are closely packed, liquids, like solids, resist compression an extremely large force is necessary to change the volume of a liquid. When a liquid is placed in a container with no lid, it remains in the container. 
That is, liquids flow (so they are a type of fluid), with the molecules held together by mutual attraction. This occurs because the atoms or molecules in a liquid are free to slide about and change neighbors. 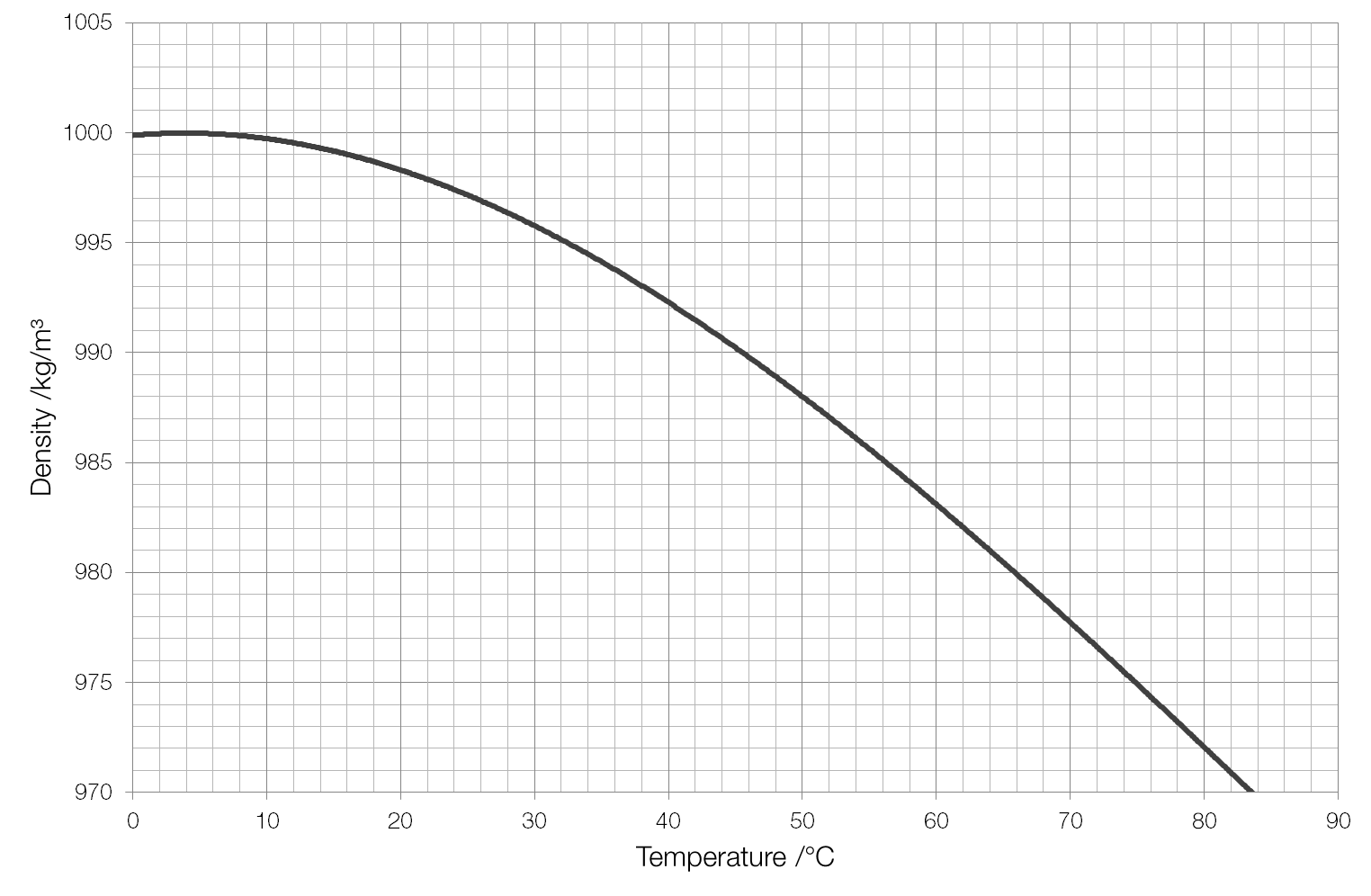
Liquids deform easily when stressed and do not spring back to their original shape once a force is removed. A gas must be held in a closed container to prevent it from expanding freely and escaping. (c) Atoms in a gas move about freely and are separated by large distances. Forces between the atoms strongly resist attempts to compress the atoms. (b) Atoms in a liquid are also in close contact but can slide over one another. Figure 14.2 (a) Atoms in a solid are always in close contact with neighboring atoms, held in place by forces represented here by springs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
